In the last 100 years antibiotics have saved millions of lives. However, they are indiscriminate weapons: they kill useful bacteria, such as those in the human gut which extract nutrients, as efficiently as they kill the nasty disease-causing sort. Ben Feringa, of the University of Groningen, in the Netherlands, and his colleagues have devised a method to make antibiotics more selective.
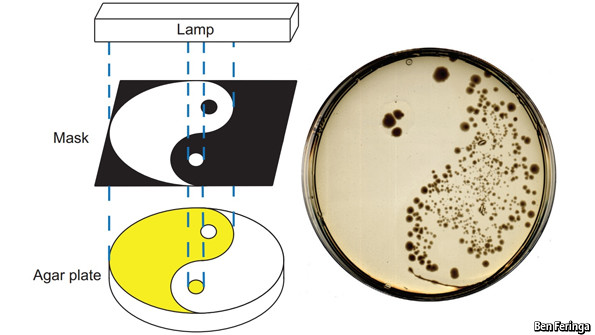
As they report in Nature Chemistry, this was achieved by slapping chemical structures called diazo compounds to a class of antibiotic called quinolones, developed to treat urinary tract infections. Different diazo compounds absorb particular wavelengths of light (it is these structures which give dyes their distinctive colour). They then added the mixture to a Petri dish containing the bacterium E. coli, shielded part of the dish and irradiated the uncovered parts with ultraviolet light. Two of the nine compounds they tested worked as hoped: whereas E. coli remained abundant in the covered parts of the Petri dish, the sections exposed to the light were almost entirely cleared of bacteria.
Crucially, in the half hour or so after exposure to the light, the diazo groups lose energy and revert to their original structure. This, in turn, switches the antibiotic off again. That way, when the antibiotic travels to other parts of the body or is excreted, it does so in its inactive form, and thus remains harmless to the friendly bacteria living in the gut. (And beyond: excreted antibiotics that make their way into sewers have been blamed for spurring the development of drug-resistant bacteria in the wild.)
The brief active window does, however, mean that Dr Feringa’s drug would only be useful in fighting localised infections, where a half-hour antibiotic raid is plenty, rather than more general ones which require a sustained onslaught. And though light can, in principle, be delivered deep inside the body using an endoscope (as happens in some cancer treatments), this is finicky and expensive in practice. As a result, light-activated antibiotics would probably be limited to easily accessible infections on the skin (in wounds, for instance) or in the mouth, ears or nose.
At least for now, that is, for Dr Feringa is working on flipping his chemical switch using sources of energy found inside the body, such as heat or certain energy-rich enzymes. If he succeeds, incidents of antibiotic friendly fire may be consigned to the history of antibacterial warfare.
First published on economist.com.
Image credit: Ben Feringa